Polonospora polonica (Błaszk.) Błaszk., Niezgoda, B.T. Goto, Magurno, comb. nov. Figure 2a–h.
MycoBank number: MB 840257; Index Fungorum number: IF 840257; Facesoffungi number: FoF;
Basionym: Acaulospora polonica Błaszk., Karstenia 27, 38. 1988.
Description: Glomerospores (= spores) formed singly in soil, arise laterally from the neck of a sporiferous saccule continuous with an extraradical mycorrhizal hypha (Fig. 2a, b). Spores hyaline to white (1A1), glistening, globose to subglobose, (80–)94(–115) μm diam, with three spore walls (Fig. 2a–h). Spore wall 1 with two layers (Fig. 2c–h). Layer 1 evanescent, hyaline, up to 1.5-μm thick, continuous with the wall of the sporiferous saccule, usually highly deteriorated or completely sloughed in mature spores. Layer 2 permanent, laminate, smooth, hyaline to white (1A1), (3.3–)4.7(–5.5) μm thick. Spore wall 2 consists of one, per- manent, flexible to semi-flexible, (0.8–)1.1(–1.3) μm thick, hyaline layer (Fig. 2c–h). Spore wall 3 composed of two permanent, hyaline, smooth layers (Fig. 2c–h). Layer 1 semi-flexible, (1.3–)1.8(–2.3) μm thick. Layer 2 flexible to semi-flexible, 0.5–1.3-μm thick, usually tightly adherent to the lower surface of layer 1 and difficult to see. In Melzer’s reagent, only spore wall 1 layer 2 sometimes stains reddish white (7A2). Germination orb not found. Sporiferous saccule hyaline, globose to subglobose, 60–90 μm diam; neck 40–70 μm long, tapering from 10.0–17.5 μm diam at the saccule to 8.0–12.5 μm diam at the point of spore attachment. Saccule wall a hyaline, smooth, 0.5–1.0-μm thick layer. Saccule collapsing at maturity and usually detached in mature spores. Cicatrix a slightly raised collar when seen in cross view, circular, 6–9 μm diam, when observed in plain view.
Mycorrhizal association: In the field, produced spores in rhizosphere of, e.g., Ammophila arenaria, Chamaecyparis lawsoniana, Corynephorus canescens, Juncus balticus, Juniperus communis, R. canina, and Thuja occidentalis (Błaszkowski 1988, 1993a, b, 1994, unpubl. data). Abundantly sporulated in trap cultures inoculated with rhizosphere soil and root fragments of R. rugosa, whose host plant was P. lanceolata. Many attempts at growing P. polonica in single-species cultures failed.
Distribution and habitat: Worldwide, with records of physical specimens and environmental sequences originating from China, Georgia, Japan, Lithuania, Poland, Portugal, and the USA (Online Resource 5). Associated with roots of plants growing in gardens, dunes, forests, and highly con- taminated sites (Gai et al. 2006; Błaszkowski 2012).
Specimens examined: Holotype. POLAND. Hel, ca 200 m from the Baltic Sea, under Thuja occidentalis, 21.08.1985, Błaszkowski J., 95, LPPDSE; 96–136 (iso-types), LPPDSE; Kuźnica, under R. rugosa, 21.08.2017, Błaszkowski J., 3810–3821, LPPDSE. LITHUANIA. Kuronian Spit, Parnidos dune, under A. arenaria, 15.09.2013, Błaszkowski J., 3804–3809, LPPDSE.
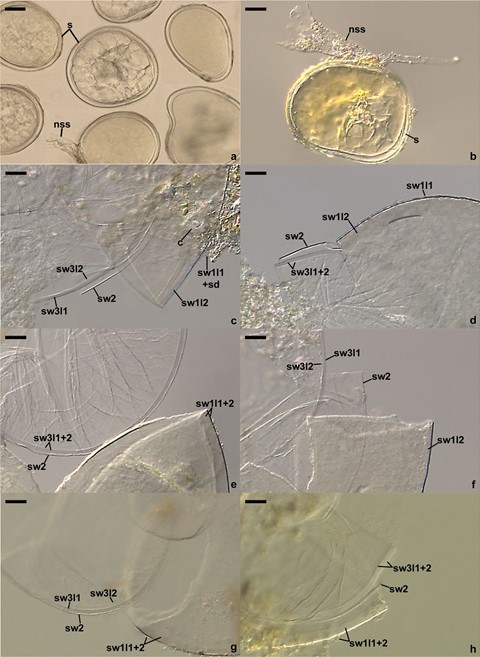
Fig. 2 Polonospora polonica. a, b. Intact spores (s) with the neck of a sporiferous saccule (nss; remnants). c‒h. Spore wall 1 (sw1) layers 1 and 2, spore wall 2 (sw2), and spore wall 3 (sw3) layers 1 and 2; soil debris (sd) covering the upper surface of sw1l1 and cicatrix
(c) are indicated in c. a‒f. Spores in PVLG. g, h. Spores in PVLG + Melzer’s reagent. a‒h. Differential interference micros- copy. Scale bars: a, b = 20 μm, c‒h = 10 μm
