Periplasma isogametum W.W. Martin & A. Warren, Mycologia 112(5): 998 (2020)
MycoBank number: MB 831250; Index Fungorum number: IF 831250; Facesoffungi number: FoF 12587;
Typification: USA. VIRGINIA: Hanover County, Ashland, unnamed tributary of Slayden Creek, small pool at the entrance to a culvert that crosses under Woodside Lane (37.767906, −77.44716), isolated in pure culture from a moribund simuliid adult, 17 Jan 2013, W. Wallace Martin WWM-203 (holotype FIG. 3F in this paper), diagnosis and description based on Pi-203-C1. Ex-holotype culture Pi-203-C1, MICH, RMC.
Etymology: isogametum, from combining forms of isos (Greek) = equal and gamia (Greek) = act of marrying. Referring to the morphology of the equal sizes and shapes of + and − gametes involved in sexual reproduction (isogamy).
Description: Mycelium compact, contorted, and holocarpic in the coelom of aquatic insects; larger, freely branched and eucarpic in culture with a prominent central vacuole, up to several cm in length, width 18–73 (¯x = 23.5 ± 12.2) μm. Thalli functioning as zoosporangia which produce short, thick-walled papillae at their tips, tip width 11.5–19 (¯x =15.7 ± 2.3) μm. In zoosporogenesis, the peripheral protoplasmic network collapses to the cell center as a series of connected zoospore initials that subsequently undergo plasmotomy and separation as individual primary zoospores. Primary zoospores slipper-shaped to broadly dumbbell-shaped, with a prolonged motile phase, transitioning directly or rounding up and undergoing a quiescent period before becoming a short- lived ellipsoidal to ovate secondary zoospore. Primary zoospores 12–19.5×4.5–6.5 (¯x = 14.8 ± 1.5 × 5.5 ± 0.5) μm, secondary zoospores 7–13 5.5–7.5 (× = 9.8 ± 1.2 × 6.5 ± 0.5) μm, whiplash flagella 18–48 (¯x = 29.9 ± 6.8) μm, tinsel flagellum 16–44 (¯x = 24.0 ± 5.8) μm. Sexual repro- duction by isogamy of rounded, thin-walled gametes: + or male gametes, 13.5–26 (¯x =18.3 ± 1.8) μm, − or female gametes, 15–32 (¯x =18.0 ± 1.8) μm, fertilization tube produced by male gamete 6.5–206 × 2.5–6 (¯x = 127.1 ± 48.2 × 4.2 ± 0.7) μm. Female gamete converted into a thick-walled oospore, typically globose to subglobose but often irregular, 21–23.5 × 19–19.5 (x= 22.3 ± 1.7 × 19.2 ± 0.2) μm, ooplast with a single eccentric oil globule.
Ecology and distribution: On moribund simuliid and chironomid adults, known only from Virginia (type), Jan to Mar. a thick-walled oospore, typically globose to subglobose but often irregular, 21–23.5 19–19.5 (¯x = 22.3 ± 1.7 × 19.2 ± 0.2) μm, ooplast with a single eccentric oil globule.
Other material examined: USA. VIRGINIA: Same location as holotype (37.767906, −77.44716), observed on several moribund simuliid adults, 17 Jan 2013, W. Wallace Martin WWM-203, prepared slides from adult simuliids (Pi-203-C1 RMC); same location as holotype, observed on several moribund simuliid adults, 11 Mar 2014, W. Wallace Martin WWM-204, prepared slides from adult simuliids and a pure culture from a single simuliid adult (Pi-204-C1 RMC); same locality as type but from a small pool at exit of culvert that crosses Providence Church Road (37.763925, −77.437234), observed on several moribund adult chironomids, 31 Mar 2015, W. Wallace Martin WWM-205, prepared slides from adult chironomids and a pure culture from a single chironomid adult.
GenBank Accession Numbers: (Pi-203-C1): 18S = MK990103; 28S = MK990102; ITS = MK990104; (Pi-205-C1 RMC): 18S = MN419333; 28S = MN410434; ITS = MN417223; cox2 = MN006990.
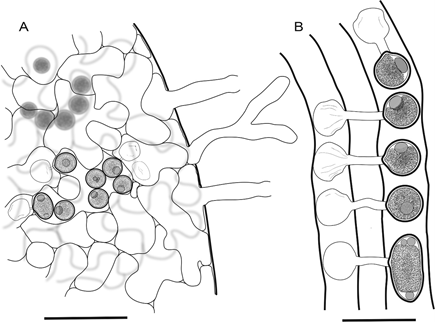
Figure 1. Periplasma isogametum. A. Mycelium in host. B. Ex-holotype Pi-203-1. Drawings of asexual and sexual stages. A. Endobiotic mycelium within host simuliid, empty zoosporangia with discharge tubes penetrating the exoskeleton and hyphae containing mature oospores. B. Conjugation of isogametes in adjacent hyphae, scalariform conjugation between four bottom pairs of gametes and lateral conjugation between uppermost gamete pair on right side. Bars: A = 80 μm; B = 40 μm.
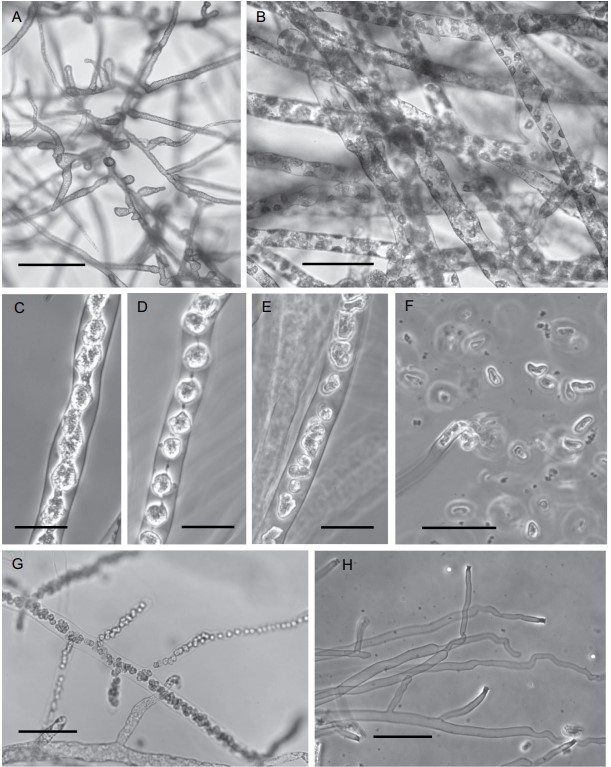
Figure 2. Periplasma isogametum (ex-holotype Pi-203-C1). Photographs of stages in vegetative and asexual reproduction. A. Vegetative mycelium in mPmTG broth. B–H. Zoosporogenesis and zoospore discharge in water. B. Initial stage of zoosporogenesis: aggregation of cytoplasm around nuclei. C. Peripheral protoplasmic network has pulled away and collapsed to the center of the zoosporangium. D. Zoospore initials united by protoplasmic strands. E. Primary zoospores swim within the zoosporangium following breaking of the protoplasmic strands. F. Primary zoospores being released following dissolution of the papilla. G. Branching zoosporangium prior to zoospore release. H. Empty zoosporangia displaying flaring thick-walled papillae. A, B, G. Bright-field images. C–F, H. Phase-contrast images. Bars: A = 150 μm; B, G, H = 50 μm; C–E = 25 μm; F = 20 μm.
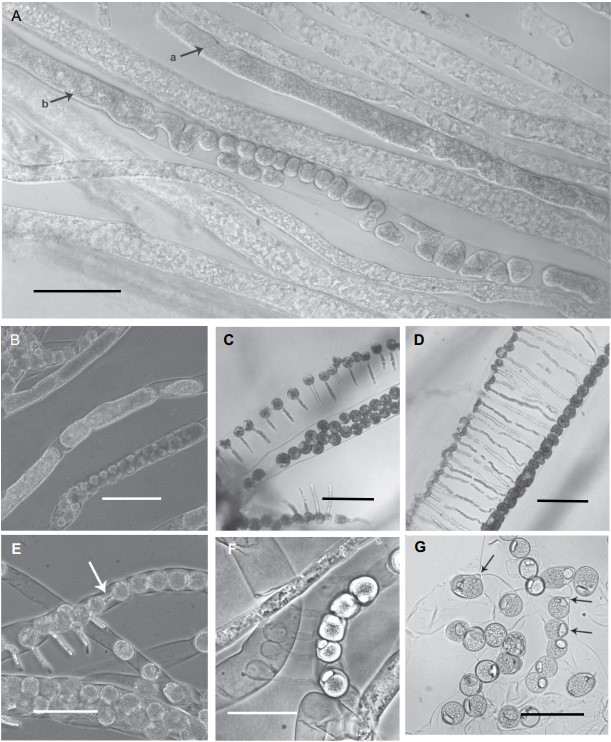
Figure 3. Periplasma isogametum (ex-holotype Pi-203-C1). Photographs of stages in sexual reproduction. A, C, D. In mPmTG agar. B, E–G. In mPmTG broth. A. Gametogenesis: a, protoplast arising from contraction, condensation, and separation of hyphal protoplasm; b, protoplast cleaving into gametes. B. Various stages in gamete development. C. Adjacent hyphae containing male gametes, with fertilization tubes (top and bottom) growing toward female gametes (center). D. Adjacent hyphae containing gametes (male to left, female to right) undergoing scalariform conjugation. E. Adjacent gametes (arrow) undergoing lateral conjugation. F. Mature oospores (right) and empty male gametes with fertilization tubes (left). G. Mature oospores with eccentric oil globules, receptive papillae on some (arrows). A, B, E, F. Phase-contrast images. C, D, G. Bright-field images. Bars: A–D = 100 μm; E, G = 75 μm; F = 50 μm.
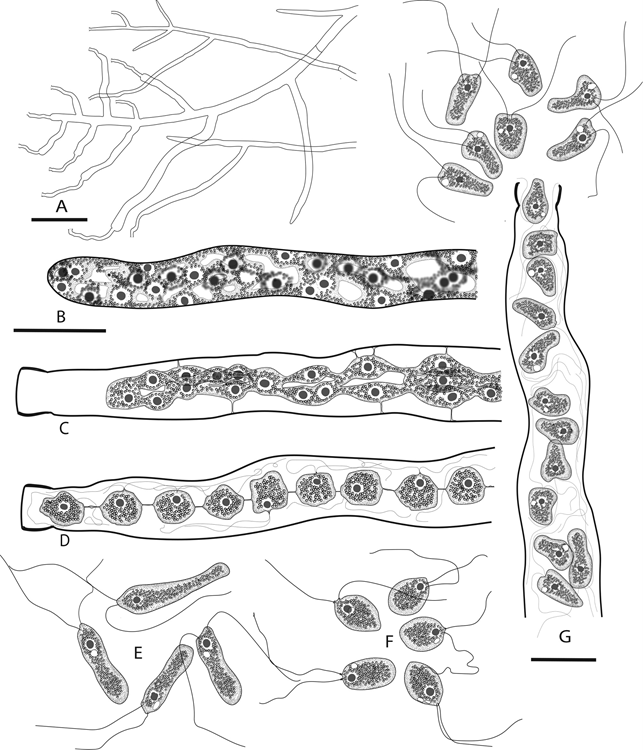
Figure 4. Periplasma isogametum (ex-holotype Pi-203-C1). Drawings of stages in zoosporogenesis, zoospore discharge, and zoospores in water. A. Branching zoosporangia following zoospore discharge. B–D, G. Sequential stages in a single zoosporangium. B. Young zoosporangium prior to papilla formation, with peripheral protoplasmic network surrounding the central vacuole. C. Protoplasmic network has collapsed to the center of the cell. D. Network is condensed into rounded zoospore initials united by protoplasmic threads. E. Primary zoospores. F. Secondary zoospores. G. Primary zoospores are released following breaking of protoplasmic threads and dissolution of the papilla tip. Bars: A = 50 μm; B (for B–F) = 15 μm; G = 15 μm.
